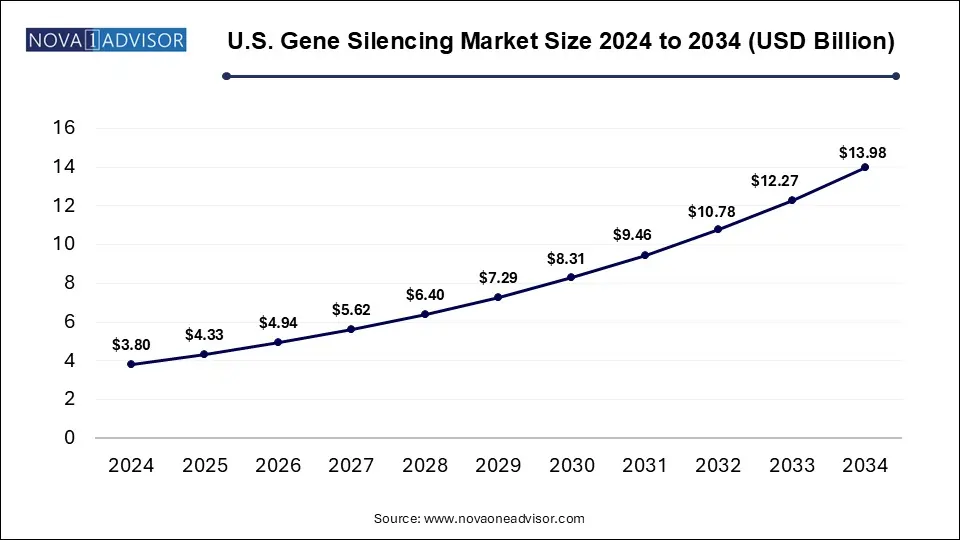
The gene silencing market accounted for USD 10.35 billion in 2024 and is expected to reach around USD 38.03 billion by 2034, growing with a CAGR of 13.9% from 2025 to 2034. This market is growing due to increasing demand for targeted therapies, advancements in RNA based technologies, and expanding applications in genetic and chronic diseases.
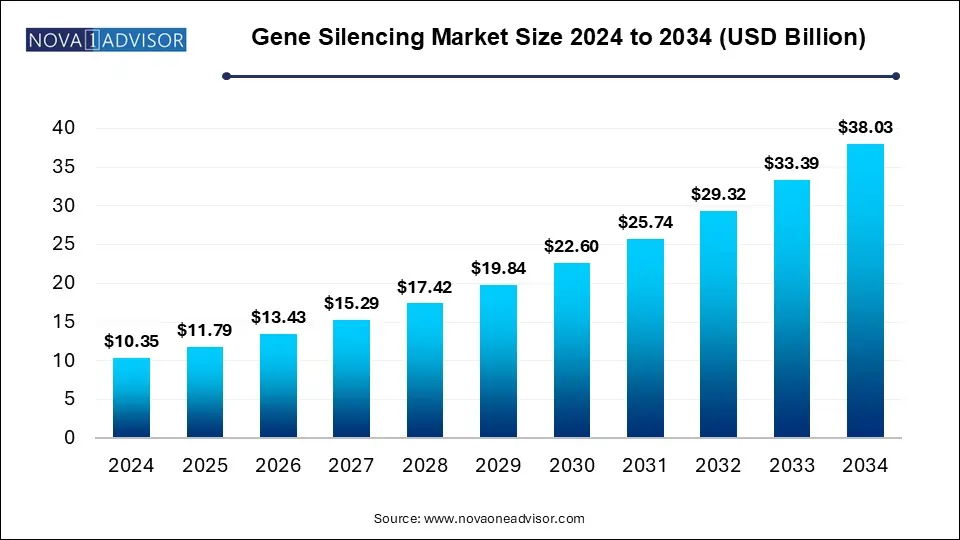
The U.S. gene silencing market size was valued at USD 3.8 billion in 2024 and is expected to reach around USD 13.98 billion by 2034, growing at a CAGR of 12.57% from 2025 to 2034.
North America dominates the gene silencing market in 2024, driven by substantial investments in RNA based therapies, a robust presence of top biotechnology and pharmaceutical firms and a well-established research infrastructure. The demand for gene silencing technologies is fueled by the region's high prevalence of genetic disorders, strong government funding and advantageous regulatory policies. North Americas market leadership is further cemented by the existence of major players like Alnylam Pharmaceuticals lonis Pharmaceutical and Arrowhead Pharmaceuticals as well as their ongoing developments in RNA Interference and antisense oligonucleotide therapies.
Asia Pacific is experiencing rapid growth in the gene silencing market driven by a growing demand for precision medicine, growing biopharmaceutical industries and rising research and development (R&D) investments. Gene silencing technology is being actively adopted by nations like China Japan and India for therapeutic and drug discovery purposes. Growing partnership between academic institutions and pharmaceutical companies' government programs to encourage biotech innovation and a quickly growing contract research and manufacturing industry are all contributing to the markets acceleration. The need for sophisticated gene therapies is also being fueled by the region's growing burden of cancer and genetic illnesses making Asia Pacific a major growth hub for a gene silencing sector.
The gene silencing market is growing due to rising demand for targeted therapies, advancements in RNA interference and antisense technologies, and increasing research in genetic disorders. Its applications in cancer, neurological diseases, and rare genetic conditions are driving the market expansion. Innovations in delivering systems and regulatory approvals for RNA based drugs further boost growth. With increasing biotech investments and industry collaborations, gene silencing continues to evolve as a key approach in precision medicine.
Market Driver
Rising Prevalence of Genetic Disorders and Chronic Diseases
The demand for gene silencing therapies which provide focused and efficient treatment options is being driven by the rising prevalence of genetic disorders, cancer and neurodegenerative disease. Since conventional therapies frequently only address symptoms rather than the underlying genetic causes, gene silencing is a desirable substitute. Interest in gene silencing strategies is further fueled by the increased awareness of precision therapeutics and personalized medicine. Gene silencing-based therapies are also becoming more and more popular among researchers and pharmaceutical companies as the prevalence of diseases like Huntington's Alzheimer's and other rare genetic disorders increases worldwide.
Increasing Demand for Personalized and Precision Medicine
Since gene silencing therapies enable highly targeted treatment approaches based on an individual's genetic profile their adoption is being driven by the shift toward personalized medicine. Gene silencing allows for precise gene regulation increasing treatment efficacy and lowering side effects in contrast to conventional therapies which frequently have broad effects. This is especially helpful for cancer and rare genetic diseases where patient outcomes can be greatly improved by tailored treatments. The development of tailored gene silencing treatments is being aided by the increasing integration of genomics bioinformation and artificial intelligence in drug development.
Strong Pipeline of Gene-Silencing Therapeutics and Clinical trials
As they advance through different phases of clinical trials an increasing number of gene silencing medications are showing encouraging outcomes in the treatment of chronic and genetic illnesses. Many pharmaceutical and biotech firms are making significant investments in the creation of drugs based on ASO and small interfering RNA and some of these have received regulatory approval. The growing clinical pipeline is boosting investor confidence and encouraging industry participants to work together strategically. The commercialization of genes is silencing market expansion as more of them obtain regulatory approvals.
| Report Coverage | Details |
| Market Size in 2025 | USD 11.79 Billion |
| Market Size by 2034 | USD 38.03 Billion |
| Growth Rate From 2025 to 2034 | CAGR of 13.9% |
| Base Year | 2024 |
| Forecast Period | 2025-2034 |
| Segments Covered | Product & Service, Application, End use, Region |
| Market Analysis (Terms Used) | Value (US$ Million/Billion) or (Volume/Units) |
| Regional scope | North America; Europe; Asia Pacific; Latin America; MEA |
| Key Companies Profiled | Revvity, Inc.; Danaher; Agilent Technologies; GenScript; Merck KGaA; Thermo Fisher Scientific, Inc.; Illumina; Bio-Rad Laboratories; Bio-Techne; New England Biolabs, Inc. |
Market opportunity
Expansion of Gene Silencing in Rare and Orphan Diseases
A promising treatment option for rare and orphan diseases which frequently have few or no effective treatment is gene silencing. Since single gene mutations are the cause of many of these disorders RNA interference and antisense oligonucleotide therapies are ideally suited to treat them. Biotech firms are concentrating more on gene silencing treatments for uncommon genetic diseases because regulatory bodies provide incentives like funding support orphan drug designations and expedited approvals. Rare disease advocacy and awareness are rising which further fosters an environment that is favorable to innovation and commercialization in the field.
Market Challenge
Regulatory and Ethical Concerns
Gene silencing regulations are still developing, and businesses have trouble getting approvals because of safety issues and the long-term effects of these treatments. To make sure gene silencing therapies don't result in immunological reactions or unanticipated genetic changes regulatory bodies need a lot of clinical data. Clear guidelines and public trust in these new therapies are also necessary because ethical concerns about gene manipulation and the possibility of unexpected consequences in human genetics continue to spark discussions.
Product segment held the largest share in 2024 stimulated by the growing use of CRISPR-based treatments antisense oligonucleotides (ASOs) and RNA interference (RNAi). To treat genetic disorders, cancers and neurodegenerative diseases pharmaceutical and biotechnology companies are making significant investments in the development of novel gene-silencing medications. The dominance of the product segment is further reinforced by the growing incidence of rare diseases and the regulatory bodies increasing approval of gene-silencing therapies. Furthermore, these treatments' market presence is increasing due to improvements in their efficiency and specificity brought about by developments in drug delivery systems like viral vectors and lipid nanoparticles.
The service segment is expected to grow at the fastest rate. driven by the growing practice of contract research organizations (CROs) and specialized biotech companies managing research and development (R&D) activities. Gene silencing services are being used by pharmaceutical companies for drug discovery target validation and personalized medicine strategies. This market is also seeing an increase in demand due to the growing need for custom oligonucleotide synthesis bioinformatics analysis and high-throughput screening. Services are a vital and quickly growing segment of the market since businesses are depending on specialized service providers for clinical trial support, regulatory consulting and manufacturing optimization as gene therapy regulations change.
Drug discovery & development segment held the largest share in 2024, the growing need for precision medicine and targeted treatments. Antisense oligonucleotides (ASOs) and RNA interference (RNAi) are two gene silencing technologies that pharmaceutical and biotech companies are using to find and validate new drug targets. These methodologies facilitate the investigation of gene functions, the screening of possible therapeutic candidates and the development of more potent therapies for cancer neurodegenerative diseases and genetic disorders. The efficiency of gene-silencing-based drug discovery is being further improved by ongoing developments in high-throughput screening and bioinformatics which is preserving this market segment's dominance.
Therapeutics segment is expected to grow at the fastest rate. driven by the growth of biopharmaceutical manufacturing and the growing commercialization of medications that silence genes. The need for large-scale economical manufacturing procedures that guarantee high purity stability and scalability is growing as more gene-silencing treatments are approved by regulators. The development of gene-silencing treatments is also being accelerated by developments in bioprocessing technologies such as lipid nanoparticle formulations RNA synthesis platforms and CRISPR-based editing. Contract manufacturing organizations (CMOs) and pharmaceutical companies are also working together to improve manufacturing processes, which is why this sector of the market is growing at one of the fastest rates.
The pharmaceutical & biotechnology segment held the largest share in 2024 driven by the increasing focus on targeted therapies and personalized medicine. Major pharmaceutical and biotech companies are heavily investing in RNA interference (RNAi), antisense oligonucleotides (ASOs) and CRISPR-based technologies to develop novel treatments for genetic disorders cancer and rare diseases. The rising number of clinical trials and regulatory approvals for gene-silencing-based drugs further solidifies this segment’s dominance. Furthermore, major industry players are strengthening their research capacities and speeding up drug development through strategic alliances mergers and acquisitions guaranteeing continued market leadership.
CROs & CMOs segment is expected to grow at the fastest rate. propelled by growing outsourcing patterns in the biotech and pharmaceutical sectors. Businesses are depending on CROs for preclinical and clinical research regulatory support and biomarker discovery as gene-silencing therapies move closer to commercialization. At the same time CMOs are essential to the expansion of RNA-based medication manufacturing gene-editing instruments and delivery systems. It is a crucial component of the growing gene-silencing landscape, and its rapid growth is being driven by the growing demand for high-quality reasonably priced manufacturing as well as developments in bioprocessing technologies.
Gene Silencing Market Recent Developments
This report forecasts revenue growth at country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2034. For this study, Nova one advisor, Inc. has segmented the Gene Silencing Market
By Product & Service
By Application
By End Use
By Regional