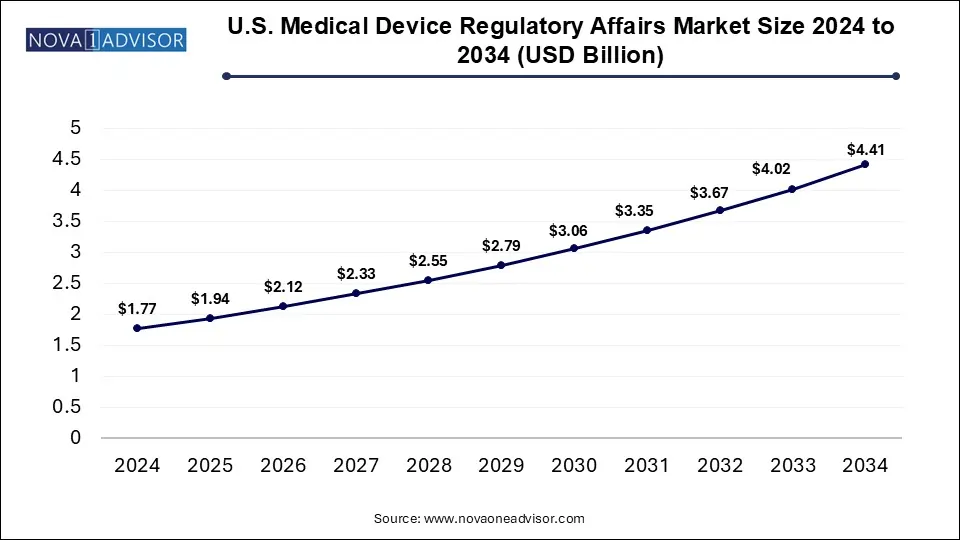
The medical device regulatory affairs market size was estimated at USD 7.15 billion in 2024 and is projected to hit around USD 17.82 billion by 2034, expanding at a CAGR of 9.56% during the forecast period from 2025 to 2034. The rising development and adoption of advanced medical devices for the treatment of a wide range of chronic diseases, such as cardiovascular, neurological disorders, cancer, and other infectious diseases are expected to boost the expansion of the medical device regulatory affairs market during the forecast period.
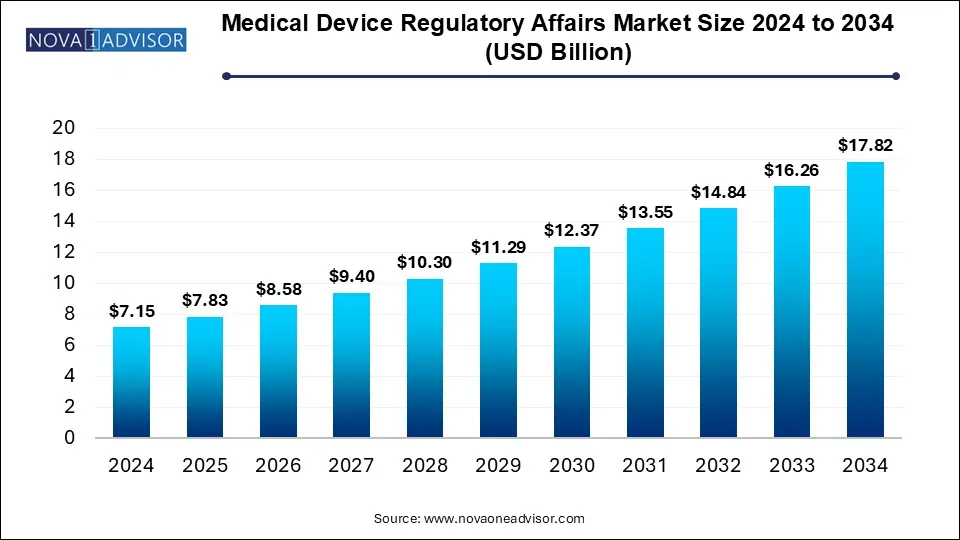
The U.S. medical device regulatory affairs market size was valued at USD 1.77 billion in 2024 and is expected to reach around USD 4.41 billion by 2034, growing at a CAGR of 8.65% from 2025 to 2034.
North America dominated the medical device regulatory affairs market. This region is home to several medical device companies increasingly outsourcing regulatory consulting and clinical trials to authorised regulatory service providers. The growing geriatric population, rising prevalence of chronic diseases among people, rapid technological innovation, and an increase in government support for the healthcare sector are expected to propel the growth of the market in the coming years. The region has the presence of the U.S. Food and Drug Administration, which regulates more than half of all medical devices worldwide. The U.S. FDA issues various guidelines for medical device manufacturers to help in the process. In addition, the surge in the number of market entrants is increasing the need for regulatory services. Such factors are contributing to the growth of the regional market.
Europe is expected to grow rapidly during the forecast period, owing to the presence of a sophisticated regulatory landscape, a growing number of clinical trials being conducted in the region, an increasing number of medical device companies, a growing burden of chronic diseases, rising technological advancement, and a supportive government framework. The region has the presence of the renowned international regulatory agencies—the European Medicines Agency (EMA), which is expected to be one of the major factors driving the market’s growth in the European region.
Medical Device regulatory affairs work to safeguard the public health by effectively controlling the efficacy and safety of products manufactured by medical device companies. Regulatory professionals ensure regulatory compliance and prepare submissions, as well as their major job function is clinical affairs or quality assurance. Medical device regulatory affairs professionals typically serve as a link between the medical device industry and regulatory bodies worldwide, including the Medicines and Healthcare Products Regulatory Agency (MHRA), United States Food and Drug Administration (USFDA), European
Medicine Agency for the European Union, Pharmaceutical & Food Safety Bureau (PFSB), Central Drugs Standard Control Organization (CDSCO), Central Drugs Standard Control Organization (CDSCO), and others.
Technological advancements in medical devices, such as AI, connected devices, software as a medical device (SaMD), machine learning, increasing trends toward portable and smaller devices, coupled with strict government regulations for medical devices are expected to accelerate the growth of the market during the forecast period.
Medical equipment regulation is a vast and rapidly changing field that is often complicated by legal challenges. Even within one regulatory framework, legal terms and their definitions are not always consistent. As technology expands, software, machine learning, and algorithms become essential components of an increasing number of digital health resources. This presents new challenges as an increasing number of instruments fall under the regulatory framework. Medical device companies have to deal with continuous changes in regulatory requirements, which can differ based on business activities and geographies. Noncompliance with the changing regulatory requirements can result in penalties and delays, which may lead to a loss of revenue. According to a survey sponsored by Genpact, 72.0% of executives from the life sciences industry consider regulatory compliance to be one of the top three challenges they face. Such factors are expected to support the demand for these services further.
| Report Coverage | Details |
| Market Size in 2025 | USD 7.83 Billion |
| Market Size by 2034 | USD 17.82 Billion |
| Growth Rate From 2025 to 2034 | CAGR of 9.56% |
| Base Year | 2024 |
| Forecast Period | 2025-2034 |
| Segments Covered | Services, Type, Service Provider, Region |
| Market Analysis (Terms Used) | Value (US$ Million/Billion) or (Volume/Units) |
| Regional scope | North America; Europe; Asia Pacific; Latin America; MEA |
| Key Companies Profiled | ICON; Plc; Emergo; Freyr; Laboratory Corporation of America Holdings; IQVIA; Inc.; Intertek Group plc; SGS Société Générale de Surveillance SA; Promedica International; Integer Holdings Corporation; Medpace |
Opportunity
Rising development of medical devices
The increasing development of medical devices by prominent medical device manufacturers is projected to offer lucrative growth opportunities to the medical device regulatory affairs market. In the healthcare industry, the regulatory function is crucial in ensuring the availability of safe healthcare products across the globe. Medical devices are the most widely used equipment in millions of lives, their utilization assists in diagnosis, prevention, and treatment of numerous diseases. Regulatory affairs play a crucial role throughout the lifecycle of a medical device as they enable the manufacturers of medical devices to devise the premarket strategy, draft regulatory submissions, and ensure post-market compliance. Therefore, thoroughly checking the quality of the medical devices is utmost importance, and the regulatory affairs experts are responsible for keeping products in conformity. Thus, bolstering the market expansion in the coming years.
Restraint
High cost and time-consuming
The high cost and time-consuming nature of regulatory compliance are anticipated to hinder the global medical device regulatory affairs market. The development, testing, and receiving approval for medical devices involves significant investments of money and long timelines, which can often result in delayed market entry, especially for potential smaller startups or companies. Such factors are likely to restrict the expansion of the medical device regulatory affairs market in the coming years.
The regulatory writing and publishing segment led the market with the highest revenue share in 2024. The large share of the segment is attributed to the fact that these services are provided from the early stages of product development to premarket approval, as high-quality documentation is essential for regulatory affairs to avoid delays in the approval process. Hence, with the increasing number of products in development, the demand for these services is likely to increase, thereby contributing to the market's growth for medical device regulatory affairs.
The segment of legal representation is anticipated to witness the fastest growth over the forecast period. This may be due to the increasing demand for legal representatives across the globe caused by the globalization of medical devices. The regulations are very complex and constantly changing. The changing regulatory landscape in regions such as Asia Pacific, MEA, and Latin America increases the demand for local experts for legal representation for obtaining regulatory approvals and customs clearance. These factors are promoting the demand for legal representation services globally.
The therapeutics segment held a maximum share in 2024. The growth of the segment is attributed to the increasing prevalence of various chronic diseases such as cancer, diabetes, cardiovascular diseases, and respiratory disorders that create demand for advanced therapeutic devices. For instance, rising demand for technologically advanced products such as auto-injectors or pen needles for effective and efficient insulin delivery in diabetic patients is fueling segment growth.
The diagnostics segment accounted for considerable growth in the global medical device regulatory affairs market over the forecast period. The high prevalence of chronic and infectious diseases is contributing to the demand for diagnosis and, thus, supporting the demand for technologically advanced diagnostic devices. Moreover, a significant number of diagnostic devices were launched in the last five years. For instance, in July 2022, Canon Medical launched the MRI machine Vantage Fortian with better scan technology and image enhancement. All these factors are expected to promote the demand for regulatory services for diagnostic devices.
The outsourcing segment registered its dominance in 2024. This is largely attributed to the lack of an in-house regulatory team. Besides, the advantages, such as cost and time savings, associated with the outsourcing of these services are contributing to market growth. Besides, new regulatory challenges, such as medical device regulation and in vitro diagnostic device regulation in Europe, as well as regulatory cybersecurity scrutiny, make market launch and recertification timelines difficult. It is incredibly challenging for businesses to manage regulatory hurdles with unskilled personnel, which drives the demand for outsourcing these services.
The in-house segment is projected to expand rapidly in the market in the coming years. Large biotechnology and medical device firms have a strong in-house team for regulatory affairs due to their varied product portfolios, pipelines, and the ability to hire skilled & experienced professionals; these are some of the key factors supporting the in-house segment. However, these firms are also shifting toward outsourcing to focus on their core competencies and address capacity issues. This is expected to reduce the demand for in-house services in the coming years.
This report forecasts revenue growth at country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2034. For this study, Nova one advisor, Inc. has segmented the Medical Device Regulatory Affairs Market
By Service
By Type
By Service Provider
By Regional